Difference between revisions of "Sandbox"
| Line 1: | Line 1: | ||
| − | * '''Description:''' | + | * '''Description:''' fructose 1,6-bisphosphate aldolase, glycolytic/ gluconeogenic enzyme<br/><br/> |
{| align="right" border="1" cellpadding="2" | {| align="right" border="1" cellpadding="2" | ||
|- | |- | ||
|style="background:#ABCDEF;" align="center"|'''Gene name''' | |style="background:#ABCDEF;" align="center"|'''Gene name''' | ||
| − | |'' | + | |''fbaA'' |
|- | |- | ||
| − | |style="background:#ABCDEF;" align="center"| '''Synonyms''' || '' | + | |style="background:#ABCDEF;" align="center"| '''Synonyms''' || ''fba, fba1, tsr '' |
|- | |- | ||
| − | |style="background:#ABCDEF;" align="center"| '''Essential''' || | + | |style="background:#ABCDEF;" align="center"| '''Essential''' || yes |
|- | |- | ||
| − | |style="background:#ABCDEF;" align="center"| '''Product''' || | + | |style="background:#ABCDEF;" align="center"| '''Product''' || fructose-1,6-bisphosphate aldolase |
|- | |- | ||
| − | |style="background:#ABCDEF;" align="center"|'''Function''' || | + | |style="background:#ABCDEF;" align="center"|'''Function''' || enzyme in glycolysis/ gluconeogenesis |
|- | |- | ||
| − | |style="background:#ABCDEF;" align="center"| '''MW, pI''' || | + | |style="background:#ABCDEF;" align="center"| '''MW, pI''' || 30,2 kDa, 5.03 |
|- | |- | ||
| − | |style="background:#ABCDEF;" align="center"| '''Gene length, protein length''' || | + | |style="background:#ABCDEF;" align="center"| '''Gene length, protein length''' || 855 bp, 285 amino acids |
|- | |- | ||
| − | |style="background:#ABCDEF;" align="center"|'''Immediate neighbours''' || ''[[ | + | |style="background:#ABCDEF;" align="center"|'''Immediate neighbours''' || ''[[spo0F]]'', ''[[ywjH]]'' |
|- | |- | ||
| − | |colspan="2" style="background:#FAF8CC;" align="center"|'''Get the DNA and protein [http://srs.ebi.ac.uk/srsbin/cgi-bin/wgetz?-e+[EMBLCDS: | + | |colspan="2" style="background:#FAF8CC;" align="center"|'''Get the DNA and protein [http://srs.ebi.ac.uk/srsbin/cgi-bin/wgetz?-e+[EMBLCDS:CAB15729]+-newId sequences] <br/> (Barbe ''et al.'', 2009)''' |
|- | |- | ||
| − | |colspan="2" | '''Genetic context''' <br/> [[Image: | + | |colspan="2" | '''Genetic context''' <br/> [[Image:fbaA_context.gif]] |
<div align="right"> <small>This image was kindly provided by [http://genolist.pasteur.fr/SubtiList/ SubtiList]</small></div> | <div align="right"> <small>This image was kindly provided by [http://genolist.pasteur.fr/SubtiList/ SubtiList]</small></div> | ||
|- | |- | ||
| Line 30: | Line 30: | ||
<br/><br/> | <br/><br/> | ||
| + | |||
=The gene= | =The gene= | ||
| Line 35: | Line 36: | ||
=== Basic information === | === Basic information === | ||
| − | * '''Locus tag:''' | + | * '''Locus tag:''' BSU37120 |
===Phenotypes of a mutant === | ===Phenotypes of a mutant === | ||
| + | |||
| + | essential [http://www.ncbi.nlm.nih.gov/pubmed/12682299 PubMed] | ||
=== Database entries === | === Database entries === | ||
| − | * '''DBTBS entry:''' [http://dbtbs.hgc.jp/COG/prom/ | + | * '''DBTBS entry:''' [http://dbtbs.hgc.jp/COG/prom/fbaA-ywjH.html] |
| − | * '''SubtiList entry:''' [http://genolist.pasteur.fr/SubtiList/genome.cgi?gene_detail+ | + | * '''SubtiList entry:''' [http://genolist.pasteur.fr/SubtiList/genome.cgi?gene_detail+BG10412] |
=== Additional information=== | === Additional information=== | ||
| − | |||
=The protein= | =The protein= | ||
| Line 52: | Line 54: | ||
=== Basic information/ Evolution === | === Basic information/ Evolution === | ||
| − | * '''Catalyzed reaction/ biological activity:''' | + | * '''Catalyzed reaction/ biological activity:''' D-fructose 1,6-bisphosphate = glycerone phosphate + D-glyceraldehyde 3-phosphate (according to Swiss-Prot) D-fructose 1,6-bisphosphate = dihydroxyacetone phosphate + D-glyceraldehyde 3-phosphate |
| − | * '''Protein family:''' | + | * '''Protein family:''' class II fructose-bisphosphate aldolase family (according to Swiss-Prot) class II fructose-bisphosphate aldolase family |
| − | * '''Paralogous protein(s):''' | + | * '''Paralogous protein(s):''' [[FbaB]] |
=== Extended information on the protein === | === Extended information on the protein === | ||
| Line 63: | Line 65: | ||
* '''Domains:''' | * '''Domains:''' | ||
| + | ** 2 x Dihydroxyacetone phosphate binding domain (210–212), (231–234) | ||
| − | * '''Modification:''' | + | * '''Modification:''' phosphorylation on Thr-212 AND Thr-234 [http://www.ncbi.nlm.nih.gov/sites/entrez/17218307 PubMed] |
| − | * '''Cofactor(s):''' | + | * '''Cofactor(s):''' 2 x zinc ion |
| − | * '''Effectors of protein activity:''' | + | * '''Effectors of protein activity:''' inhibited by alpha-keto acids [http://www.ncbi.nlm.nih.gov/pubmed/24624 PubMed] |
* '''Interactions:''' | * '''Interactions:''' | ||
| Line 78: | Line 81: | ||
* '''Structure:''' | * '''Structure:''' | ||
| − | * '''Swiss prot entry:''' [http://www.uniprot.org/uniprot/ | + | * '''Swiss prot entry:''' [http://www.uniprot.org/uniprot/P13243 P13243] |
| − | * '''KEGG entry:''' [http://www.genome.jp/dbget-bin/www_bget?bsu+ | + | * '''KEGG entry:''' [http://www.genome.jp/dbget-bin/www_bget?bsu+BSU37120] |
| − | * '''E.C. number:''' | + | * '''E.C. number:''' [http://www.expasy.org/enzyme/4.1.2.13 4.1.2.13] 3 4.1.2.13] |
=== Additional information=== | === Additional information=== | ||
| + | |||
| + | Binds 2 zinc ions per subunit. One is catalytic and the other provides a structural contribution | ||
=Expression and regulation= | =Expression and regulation= | ||
* '''Operon:''' | * '''Operon:''' | ||
| + | **''fbaA'' [http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Abstract&list_uids=+11489127 PubMed] | ||
| + | **''fbaA-[[ywjH]]'' [http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Abstract&list_uids=+11489127 PubMed] | ||
| − | * ''' | + | * '''Sigma factor:''' |
| − | * '''Regulation:''' | + | * '''Regulation:''' constitutively expressed [http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Abstract&list_uids=+11489127 PubMed] |
* '''Regulatory mechanism:''' | * '''Regulatory mechanism:''' | ||
| − | * '''Additional information:''' | + | * '''Additional information:''' |
=Biological materials = | =Biological materials = | ||
| − | * '''Mutant:''' | + | * '''Mutant:''' |
| − | * '''Expression vector:''' | + | * '''Expression vector:''' pGP395 (N-terminal His-tag, in [[pWH844]]), pGP88 (N-terminal Strep-tag, for [[SPINE]], expression in ''B. subtilis'', in [[pGP380]]) |
| − | + | ||
| − | * '''lacZ fusion:''' | + | * '''lacZ fusion:''' pGP601 (in [[pAC6]]) |
* '''GFP fusion:''' | * '''GFP fusion:''' | ||
| − | * '''two-hybrid system:''' | + | * '''two-hybrid system:''' ''B. pertussis'' adenylate cyclase-based bacterial two hybrid system ([[BACTH]]), available in [[Stülke]] lab |
* '''Antibody:''' | * '''Antibody:''' | ||
| Line 118: | Line 125: | ||
=References= | =References= | ||
| − | <pubmed> | + | <pubmed>17218307, </pubmed> |
| − | # | + | # Trach K, Chapman JW & Piggot P (1988) Complete sequence and transcriptional analysis of the ''spo0F'' region of the ''Bacillus subtilis'' chromosome ''J Bacteriol.'' '''170:''' 4194-4208. [http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Abstract&list_uids=+2457578 PubMed] |
| − | # | + | # Ludwig H, Homuth G & Schmalisch M (2001) Transcription of glycolytic genes and operons in ''Bacillus subtilis'': evidence for the presence of multiple levels of control of the ''gapA'' operon ''Mol Microbiol.'' '''41:''' 409-422. [http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Abstract&list_uids=+11489127 PubMed] |
| + | # Macek et al. (2007) The serine/ threonine/ tyrosine phosphoproteome of the model bacterium ''Bacillus subtilis''. Mol. Cell. Proteomics 6: 697-707 [http://www.ncbi.nlm.nih.gov/pubmed/17218307 PubMed] | ||
Revision as of 13:16, 8 June 2009
- Description: fructose 1,6-bisphosphate aldolase, glycolytic/ gluconeogenic enzyme
| Gene name | fbaA |
| Synonyms | fba, fba1, tsr |
| Essential | yes |
| Product | fructose-1,6-bisphosphate aldolase |
| Function | enzyme in glycolysis/ gluconeogenesis |
| MW, pI | 30,2 kDa, 5.03 |
| Gene length, protein length | 855 bp, 285 amino acids |
| Immediate neighbours | spo0F, ywjH |
| Get the DNA and protein sequences (Barbe et al., 2009) | |
Genetic context 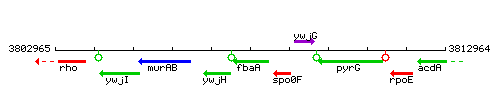
This image was kindly provided by SubtiList
| |
Contents
The gene
Basic information
- Locus tag: BSU37120
Phenotypes of a mutant
essential PubMed
Database entries
- DBTBS entry: [1]
- SubtiList entry: [2]
Additional information
The protein
Basic information/ Evolution
- Catalyzed reaction/ biological activity: D-fructose 1,6-bisphosphate = glycerone phosphate + D-glyceraldehyde 3-phosphate (according to Swiss-Prot) D-fructose 1,6-bisphosphate = dihydroxyacetone phosphate + D-glyceraldehyde 3-phosphate
- Protein family: class II fructose-bisphosphate aldolase family (according to Swiss-Prot) class II fructose-bisphosphate aldolase family
- Paralogous protein(s): FbaB
Extended information on the protein
- Kinetic information:
- Domains:
- 2 x Dihydroxyacetone phosphate binding domain (210–212), (231–234)
- Modification: phosphorylation on Thr-212 AND Thr-234 PubMed
- Cofactor(s): 2 x zinc ion
- Effectors of protein activity: inhibited by alpha-keto acids PubMed
- Interactions:
- Localization:
Database entries
- Structure:
- Swiss prot entry: P13243
- KEGG entry: [3]
- E.C. number: 4.1.2.13 3 4.1.2.13]
Additional information
Binds 2 zinc ions per subunit. One is catalytic and the other provides a structural contribution
Expression and regulation
- Sigma factor:
- Regulation: constitutively expressed PubMed
- Regulatory mechanism:
- Additional information:
Biological materials
- Mutant:
- Expression vector: pGP395 (N-terminal His-tag, in pWH844), pGP88 (N-terminal Strep-tag, for SPINE, expression in B. subtilis, in pGP380)
- lacZ fusion: pGP601 (in pAC6)
- GFP fusion:
- two-hybrid system: B. pertussis adenylate cyclase-based bacterial two hybrid system (BACTH), available in Stülke lab
- Antibody:
Labs working on this gene/protein
Your additional remarks
References
- Trach K, Chapman JW & Piggot P (1988) Complete sequence and transcriptional analysis of the spo0F region of the Bacillus subtilis chromosome J Bacteriol. 170: 4194-4208. PubMed
- Ludwig H, Homuth G & Schmalisch M (2001) Transcription of glycolytic genes and operons in Bacillus subtilis: evidence for the presence of multiple levels of control of the gapA operon Mol Microbiol. 41: 409-422. PubMed
- Macek et al. (2007) The serine/ threonine/ tyrosine phosphoproteome of the model bacterium Bacillus subtilis. Mol. Cell. Proteomics 6: 697-707 PubMed